- Clinical Data as a Service (CDaaS) is Caresyntax’s suite of tech and professional services to collect and analyze real world evidence (RWE) generated in the normal course of treatment throughout a surgical patient process.
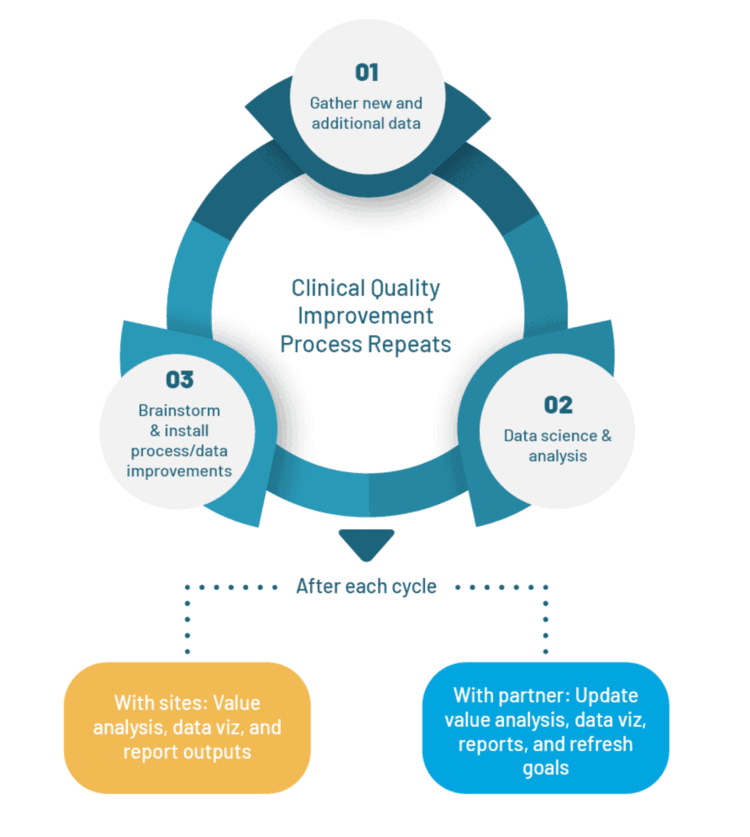
- CDaaS follows a Clinical Quality Improvement (CQI) methodology to gather data and provide analyses to drive value–based quality improvement initiatives.
- RWE captured in these projects can be collected without requiring Institutional Review Board (IRB) submission or individual patient consent (as discussed in more detail below). Clinical Quality Improvement initiatives do not meet the criteria for research on human subjects according to the US Department of Health and Human Services.
- Industry partners can use CDaaS to analyze de-identified RWE and RWD about the use of their product(s) in operating rooms in the US, Europe, and beyond. Caresyntax acts as a data analytics engine for both clinical and industry partners, and takes on the burden of data collection, cleansing, analysis, and reporting. The outputs of a CDaaS project are discussed in more detail below, but can be any form of data analytics, reports, white papers, presentations, publications, regulatory submissions, etc., based on partner needs.
- The CDaaS CQI method is FDA-validated when conducted as part of a CQI initiative in partnership with clinical teams and healthcare providers.
- A CDaaS project can capture and analyze any data generated throughout the entire surgical patient journey. This could include data points from the pre– and post-operative settings, along with any data used or generated during surgery.
- In terms of data modalities, a CDaaS project could include clinical data from:
- Electronic medical records (EMRs)
- Imaging data (including diagnostic imaging and scans)
- Video (including endoscopic and in-room cameras)
- Medical device data
- Direct patient follow-up including long-term outcomes information (PREMs. PROMs, patient-facing portals and apps)
- Hospital financial data
- This is a non-exhaustive list of data modalities – datasets are customized to fit the needs of each project
- The specific data points to be collected are determined in the initial CDaaS workshops, aligning around the needs of the industry partner and capabilities of the participating clinical sites.
- The data collection process is not limited to a prescribed protocol – it is continuously adapted throughout the project, giving the industry and clinical teams the opportunity to adjust their datasets and methodologies in response to new learnings and CQI initiatives.
- CDaaS projects produce two categories of outputs: CQI deliverables to the clinical teams, and de–identified commercial analyses and reports to any involved industry partners
- For clinicians/providers: CQI deliverables are a series of impartial, value-based assessments reporting on the entire patient process for a specified surgical procedure. Data is collected and analyzed in three–month cycles referred to as “CQI cycles”. A deliverable is generated at the conclusion of each CQI cycle, and usually includes a combination of pre-, peri-, and post-operative data, EMR/EHR information, outcomes, PREMs/PROMs, financial, and efficiency analyses, depending on the specifics of the project. The deliverables are shared with the surgical care team and other relevant stakeholders in the form of reports and sophisticated data visualization dashboards.
- For industry partners: de–identified commercial analyses are customized to the needs of a specific project. They are generally designed to improve patient outcomes and to document the value story of the product being evaluated in the CQI project. CDaaS methodology is uniquely able to identify the optimal subpopulations and techniques which maximize the value of a given product. Previous CDaaS outputs have taken the form of sales collateral, abstracts, and manuscripts, and CDaaS data has been utilized in submissions which resulted in multiple successful FDA awards.
- Based on our expertise with clinical data ingestion, curation, data science, and quality improvement for value–based care, many clinical teams engage with Caresyntax to perform CQI projects directly on their behalf.
- For projects with an industry sponsor, Caresyntax engages with clinical partners (including those suggested by the sponsor’s team) to identify surgeons whose practice best meets the criteria of the proposed project. Caresyntax contracts with those clinical teams (and their related stakeholders as necessary) to allow sharing of data and to provide them with impartial CQI deliverables regarding the procedure in question.
- The methodology has been reviewed and approved by an independent ethics board, and the FDA has repeatedly awarded product indications and labeling based on data collected through CDaaS projects. To date, Caresyntax partners have included CDaaS data as part of multiple FDA submissions and numerous peer-reviewed journals and conferences.
- Based on our expertise with clinical data ingestion, curation, data science, and quality improvement for value-based care, many clinical sites engage with Caresyntax to perform CQI projects directly on their behalf.
- For projects with an industry sponsor, Caresyntax engages with clinical sites (including those suggested by the sponsor’s team) to identify surgeons whose practice best meets the criteria of the proposed project. Caresyntax contracts with those clinical teams
- As the intermediary party between clinical sites and industry partners, the raw clinical data always belongs to the clinical site where it was collected. Caresyntax, through the data collection and curation process, creates a set of de-identified data records, analyses, and reports for the project. The final de-identified dataset and analytics are delivered to the industry partner, with full ownership rights for publication, commercial use, etc.
- Yes – CDaaS data has already been included in numerous peer-reviewed publications. Due to the speed of our process, we can work with partners to scope a project with very specific timelines – for instance, many of our partners will launch a CDaaS project with a plan to have data available for submission to upcoming conferences.
- Caresyntax has partner sites in both the US and the EU that can be included for a clinical data project. However, CDaaS projects are not limited to current Caresyntax sites – we can deploy our data collection system and methodology anywhere the partner’s product (device, drug, technology, or service) is in use today.
- We have extensive experience with GDPR and HIPAA regulations, and we work with our partners on a per-project basis to understand the regulatory requirements of the project, and to ensure that all data processing and storage is handled in compliance with all applicable regulations.
- Not necessarily. In most cases, a CDaaS project does not require IRB review or approval because the project does not meet the criteria for research on human subjects as outlined by the HHS/FDA. Because of our partnership directly with the surgical teams in a healthcare operations role for quality improvement, we can provide de–identified analyses to industry partners rapidly.
- The methodology for CDaaS has been reviewed and approved by an independent ethics board. If, for a specific project, oversight by an IRB or other Ethics Board is preferred by the industry partner or one of the participating clinical sites, Caresyntax can facilitate the deployment of a CDaaS project with central and/or local IRBs (often under an IRB-approved waiver of consent).
- CDaaS projects are much faster than traditional clinical trials for three key reasons:
- CDaaS projects capture RWE generated in the standard of care setting and don’t rely on data entry into an electronic data capture system (EDC)
- CDaaS projects generally don’t require IRB or Ethics Board review (and if they do require IRB review, it is generally under a waiver of consent)
- CDaaS projects can drive results with relatively small datasets: most of our projects only require several dozen to a few hundred patients to identify meaningful levers of value and potential quality improvement opportunities.
- These factors allow us to scope a project, collect, cleanse, and analyze data, and deliver actionable insights in months instead of the years required for a traditional clinical trial.